ACID RAIN
Acid Rain: How It Works ??
If you hike through the Appalachian Mountains, you'll
spot stands of dead and weakened trees. If you live in a city, you might notice
worn stone buildings, streaks on your car roof or corroded metal railings and
statues. You can see the effects of acid rain nearly everywhere you go, but with media and
public attention turned to the more ominous prospect of global warming, acid rain has
fallen by the wayside. The scourge from the sky almost seems like a
20th-century problem -- an issue dealt with in the 1980s and 1990s by
legislation.
Acid rain occurs mostly in the Northern Hemisphere -- the more industrialized, dirtier half of the globe. Winds can sweep up emissions from high smokestacks and carry pollutants far from their original sources, crossing state lines and national borders in the process. Acid rain may not have the complete global range of greenhouse gases, but it is a transboundary, and therefore international, issue.
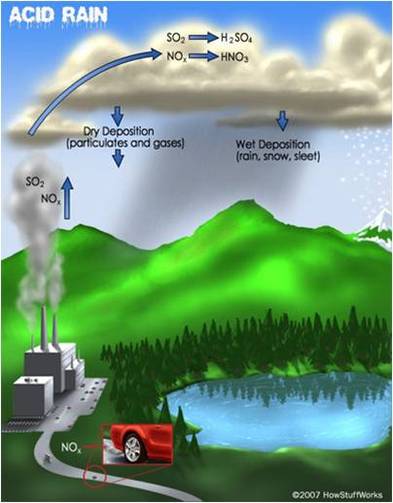
Acid rain, also known as acid deposition, is caused by emissions of sulfurdioxide (SO2) and nitrogen oxides (NOx) from power plants, cars and factories. Natural sources like volcanoes, forest fires and lightning strikes also add to the man-made pollution. SO2 and NOx become acids when they enter the atmosphere and react with water vapor. The resulting sulfuric and nitric acids can fall as wet or dry depositions. Wet deposition is precipitation: acid rain, snow, sleet or fog. Dry deposition falls as acidic particulates or gases.
Acid rain occurs mostly in the Northern Hemisphere -- the more industrialized, dirtier half of the globe. Winds can sweep up emissions from high smokestacks and carry pollutants far from their original sources, crossing state lines and national borders in the process. Acid rain may not have the complete global range of greenhouse gases, but it is a transboundary, and therefore international, issue.
Acid rain, also known as acid deposition, is caused by emissions of sulfurdioxide (SO2) and nitrogen oxides (NOx) from power plants, cars and factories. Natural sources like volcanoes, forest fires and lightning strikes also add to the man-made pollution. SO2 and NOx become acids when they enter the atmosphere and react with water vapor. The resulting sulfuric and nitric acids can fall as wet or dry depositions. Wet deposition is precipitation: acid rain, snow, sleet or fog. Dry deposition falls as acidic particulates or gases.
The pH of Acid Rain
Scientists express
the acidity of acid rain using the pH scale. The scale defines a solution's acidity, neutrality or
alkalinity based on its concentration of hydrogen ions. Acids have a high
concentration of hydrogen ions and a low pH.
The scale ranges from zero to 14, with pure water at a neutral 7.0. Most water,
however, is not exactly pure. Even clean, normal rain has a pH of about 5.6.
This is because it reacts with carbon dioxide in the atmosphere and forms
mildly acidic carbonic acid before it becomes rain.
Acid rain has a pH of 5.0 or less. Most acid deposition ranges from pH 4.3 to 5.0 -- somewhere between the acidity of orange juice and black coffee. But comparing acid rain to safe, natural acids can be misleading. Even at its weakest, acid rain wrecks ecosystems by stunting sensitive plants and killing delicate aquatic eggs. |
The Effects Of Acid Rain
Forests rely on their
soil's buffering capacity to protect them from acid rain. Acidic waters draw
out soil toxins like aluminum. Trees take in the poisonous substances, and
runoff dumps it in lakes, rivers and streams. Acid rain also dissolves helpful
minerals and nutrients like calcium, magnesium and potassium before trees can
absorb them. Acid rain rarely kills a forest outright but instead stunts its
growth through years of soil degradation. Nutrient deprivation and exposure to
toxins make trees more likely to topple in storms or die in cold weather.
Even trees in well-buffered soil can weaken in harsh acid fog. High-elevation forests soak in acidic clouds, which strip leaves of nutrients and break down trees' ability to resist cold. The bald peaks of the Appalachian Mountains tell of the poisonous effect of acid rain on high-elevation forests.
Even trees in well-buffered soil can weaken in harsh acid fog. High-elevation forests soak in acidic clouds, which strip leaves of nutrients and break down trees' ability to resist cold. The bald peaks of the Appalachian Mountains tell of the poisonous effect of acid rain on high-elevation forests.
Acid Rain and the Taj Mahal

Taj Mahal
Damage from acid
rain mars some of the world’s finest cultural monuments. Emissions reductions,
however, have helped slow the rate of damage in North America and Europe.
India’s Taj Mahal has not been spared as well. The mausoleum built by the Mughal Emperor Shah Jahan for his beloved wife Mumtaz Mahal is losing its white luster and turning a sickly pale shade.
Scientists blame pollution from local foundries and a nearby oil
refinery for this plight.
Like the Taj Mahal, many monuments face danger, come lets join hands and eliminate the pollution danger !!
Like the Taj Mahal, many monuments face danger, come lets join hands and eliminate the pollution danger !!